Is Insulin Resistance Quietly Driving Your Chronic Pain?
- Jason Winkelmann
- Apr 8
- 11 min read

TL;DR
Insulin resistance is not just a blood sugar problem. It is a systems-level disruption that directly affects inflammation, nerve function, and pain processing. It can amplify chronic pain even in people without diabetes. By altering how your body handles glucose, regulates inflammation, and modulates nerve signaling, insulin resistance creates a self-perpetuating cycle of pain and physiological stress. Addressing it requires more than symptom control. It requires restoring metabolic and nervous system balance.
What Patients Are Usually Told
Most patients are told that insulin resistance is primarily a concern for people with diabetes or prediabetes. It’s framed as a blood sugar issue; something that matters for long-term complications like cardiovascular disease or nerve damage years down the line.
On the surface, this explanation sounds logical. Blood sugar regulation is clearly important, and insulin plays a central role in that process.
But this is where the explanation becomes incomplete.
What many patients are never told is that insulin is not just a “blood sugar hormone.” It is a signaling molecule that directly influences inflammation, nervous system function, tissue repair, and pain processing in real time. Not just decades later.
Where That Explanation Breaks Down
If insulin resistance only mattered for diabetes, it would not show up so consistently across nearly every chronic pain condition.
Yet it does.
Patients with chronic low back pain, fibromyalgia, migraines, neuropathy, and autoimmune conditions frequently share one common thread: dysregulated inflammation and metabolic dysfunction.
The problem with the conventional explanation is that it isolates insulin resistance to one system (blood sugar), when, in reality, it is affecting multiple systems simultaneously.
Symptoms Are Signals, Not the Problem
Pain is not the problem. It is the output.
This is a core principle in chronic pain physiology: symptoms are signals generated by systems under stress. Not the root cause themselves .
When inflammation is dysregulated, when nerves become hypersensitive, when energy production is impaired, the brain produces pain.
What this means for patients is that reducing pain without addressing the systems producing it will always be incomplete.
The Physiology Most Patients Are Never Taught
Normal Function: How Insulin Is Supposed to Work
When you eat carbohydrates, they are broken down into glucose in your bloodstream.
Your body does not want glucose to remain in the blood. It needs to enter your cells to be used for energy.
Insulin acts like a key. It binds to receptors on the cell, opening the door so glucose can move inside.
Inside the cell, that glucose is used to produce energy, support cellular repair, and maintain normal function.

What Changes in Insulin Resistance
Over time, with repeated high glucose exposure, cells become overloaded.
Instead of accepting more glucose, they begin to resist insulin’s signal.
Now the body compensates by producing more insulin to force glucose into the cell. Eventually:
Cells stop responding effectively to insulin
Glucose builds up in the bloodstream
Insulin levels remain chronically elevated
This is insulin resistance.
How Insulin Resistance Produces Chronic Pain
This is the part most patients are never taught.
They may be told insulin resistance is “bad for inflammation” or “something to watch so you do not become diabetic.” But that framing is far too small. Insulin resistance does not simply raise blood sugar on a lab report. It changes the biochemical environment in which your nerves, immune cells, spinal cord, and brain function every day.
What this means for patients is that insulin resistance is not sitting quietly in the background. It is actively changing how pain is created, amplified, repaired, and interpreted.
Inflammation Stops Being Protective and Starts Becoming Self-Sustaining
Inflammation is not inherently bad. Under normal circumstances, it is one of the body’s most important repair tools. If you sprain an ankle, get an infection, or injure tissue, inflammatory signals help recruit immune cells, clear debris, and coordinate healing.
That is normal inflammation.
The problem begins when inflammation stops behaving like a short-term repair response and becomes a chronic biochemical state.
This is where insulin resistance becomes so important.
When glucose remains elevated in the bloodstream because cells are not responding appropriately to insulin, that excess glucose does not just “sit there.” It begins activating inflammatory pathways. One of the major ones is NF-kappa B, which acts like a master switch for chronic inflammation. Once that pathway is turned on, it stimulates the production of inflammatory cytokines such as TNF-alpha, IL-1, and IL-6. These are not abstract lab markers. These are chemical messengers that directly change how tissues and nerves behave.

They increase immune activation. They increase oxidative stress. They sensitize nociceptors, which are the nerves responsible for detecting threat and transmitting pain signals.
This is where the explanation becomes incomplete in conventional care. Patients are often told they are inflamed, but not why that inflammatory state keeps returning. The missing piece is that insulin resistance helps create the metabolic conditions that keep re-triggering inflammatory signaling.
In simpler terms, insulin resistance makes it easier for the body to stay stuck in “alarm mode.”
That matters because once nociceptors are repeatedly exposed to inflammatory chemicals, they become easier to activate. Signals that once required a meaningful injury can now be triggered by much less. The threshold drops. Pain becomes easier to produce.
What this means for patients is that inflammation is no longer just a response to damage. It becomes part of the machinery generating the pain itself.
Insulin Resistance Creates a Feed-Forward Loop That Traps the Body in Pain
One of the biggest reasons chronic pain becomes chronic is that it stops behaving like a straight line.
Most people imagine pain like this: injury leads to inflammation, inflammation leads to pain, pain eventually goes away.
But chronic pain rarely works that cleanly. It behaves more like a loop.
Insulin resistance is one of the reasons that loop becomes so difficult to break.
Here is what happens.
When insulin resistance keeps glucose elevated, inflammatory pathways are activated. Those pathways sensitize nerves and increase pain. But pain itself is a physiological stressor. When the brain perceives pain as an ongoing threat, it activates the stress response through the sympathetic nervous system and the HPA axis. That means more adrenaline, more cortisol, more vigilance.
Cortisol is often discussed as a “stress hormone,” but one of its major jobs is to increase fuel availability. In practical terms, that often means raising blood glucose.
So now the body is doing something very unfortunate but very logical:
insulin resistance raises blood glucose
elevated glucose increases inflammatory signaling
inflammation increases pain
pain activates the stress response
stress hormones raise glucose even more
higher glucose worsens insulin resistance
Now the cycle repeats.
This is why many patients feel like their body is stuck in a pattern they cannot get out of. Because in many cases, it is.
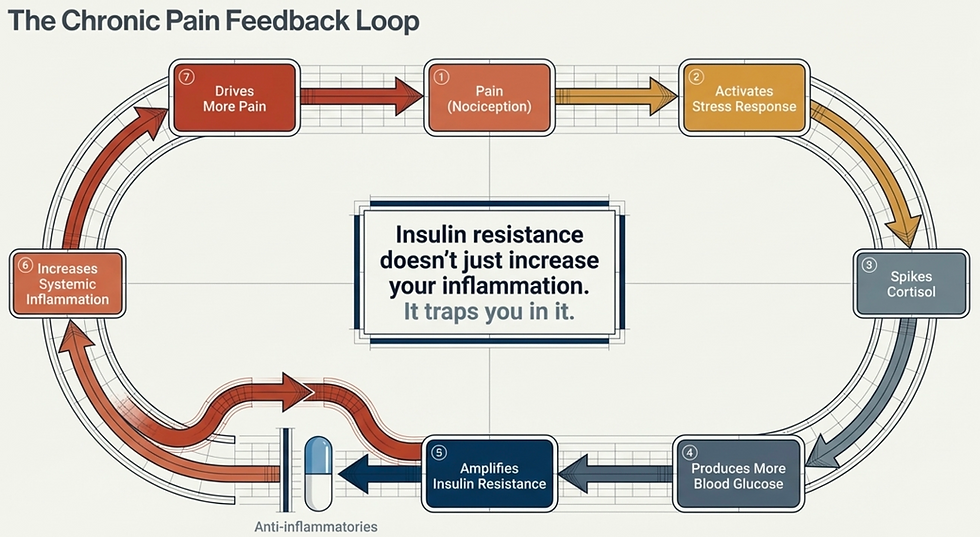
The problem with oversimplified pain models is that they treat inflammation, stress, blood sugar, and pain as separate topics. They are not separate topics. In chronic illness, they are part of the same network.
What this means for patients is that every flare is not just “more pain.” It may also be more stress chemistry, more glucose dysregulation, more inflammatory signaling, and less capacity to calm the system back down.
This also explains why symptom suppression alone often loses effectiveness over time.
If you only block one piece of the loop while the rest of the loop remains active, the system keeps rebuilding the same problem.
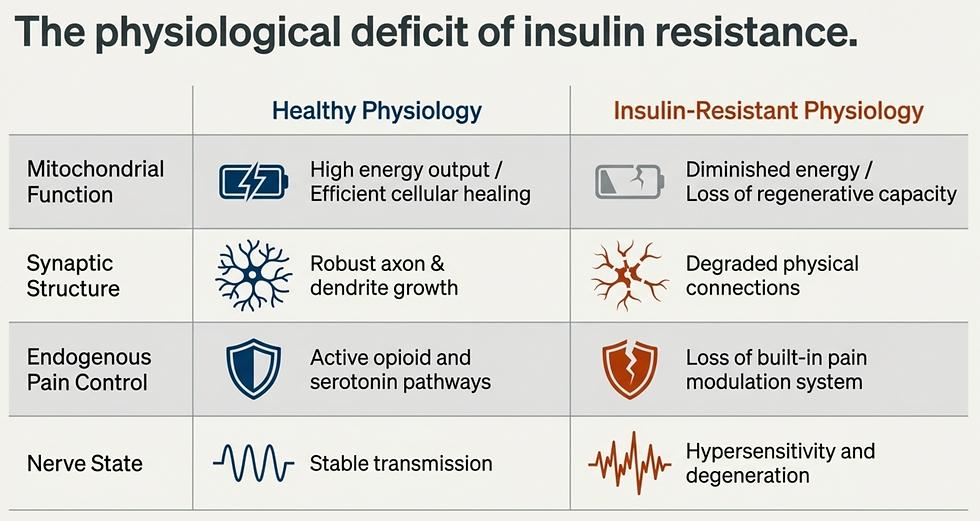
Nerve Repair and Nerve Energy Production Begin to Break Down
Patients are often told that insulin resistance is about sugar, weight, or future diabetes risk. What many are never told is that insulin is also a growth and survival signal for the nervous system.
Nerves do not just need protection from damage. They need constant support to maintain themselves, communicate properly, and repair after injury or inflammation.
Under healthy conditions, insulin helps neurons do several critical things:
That matters because neurons are metabolically expensive cells. They require enormous amounts of energy to maintain balance, send signals, repair membranes, recycle neurotransmitters, and adapt to changing demands. They cannot do this well in an unstable metabolic environment.
With insulin resistance, that support weakens:
The neuron becomes less efficient at using fuel.
Mitochondria begin producing energy less effectively.
Oxidative stress rises.
Repair capacity drops.
Regeneration slows.
Communication between neurons becomes less coordinated and more fragile.
This is especially important in sensory pathways, where healthy nerve function depends on the ability to accurately detect, transmit, and regulate incoming information.
When that system loses metabolic support, two things often happen at once:
the nerve becomes worse at repairing itself
the nerve becomes more likely to misfire or amplify threat signals
That combination is a major driver of chronic pain.
This is also why nerve-related symptoms often feel confusing. Patients may experience burning, buzzing, tingling, stabbing, electrical pain, hypersensitivity, or pain that seems disproportionate to any visible injury. They are often told nothing serious is wrong because imaging does not explain it.
But nerves do not need a herniated disc or severe structural compression to malfunction. They can become metabolically stressed, inflamed, undernourished, and hyperexcitable.
What this means for patients is that insulin resistance may be quietly taking away the nervous system’s ability to calm, repair, and regulate itself, even before diabetes is ever diagnosed.

The Spinal Cord Becomes More Permissive to Pain
Most people think pain is created where the body hurts.
That is only partially true.
Pain begins with signals in the body, but those signals are filtered and modified at multiple levels before they ever reach the brain and become a conscious experience. One of the most important filtering centers is the spinal cord, particularly the dorsal horn.
Under normal conditions, not every signal coming from the body is allowed through. That would be overwhelming. The nervous system constantly decides what is important and what is background noise. Some signals are amplified. Others are suppressed.
This is part of what patients hear described as descending inhibition. The brain and brainstem sending inhibitory signals downward to help limit how much nociceptive traffic gets through.
When this system is working well, minor inputs stay minor. Pressure feels like pressure. Clothing feels like clothing. Movement feels like movement.
But with chronic pain, this filtering system becomes less effective. More signals get through. Signals are amplified more easily. Inputs that should be interpreted as neutral begin to feel painful.
And insulin resistance appears to contribute to that process.
Insulin receptors are present in the central nervous system, including areas involved in pain regulation. When insulin signaling is impaired, the spinal cord becomes more vulnerable to neuroinflammation, reduced inhibition, and abnormal sensory gating.
Inflammatory cytokines in the central nervous system change how neurons communicate. Microglia and astrocytes become more reactive. Excitatory signaling increases. Inhibitory control weakens.
This creates a very different environment inside the dorsal horn.

Instead of a calm filtering station, it becomes a more excitable relay center. That means more pain signals pass upward. It also means the threshold for pain drops. Over time, the nervous system becomes more practiced at producing pain and less practiced at suppressing it.
What this means for patients is that chronic pain is not just about having more input from the body. It is also about losing the spinal cord’s ability to appropriately regulate what gets through.
This is why people with chronic pain often say things like:
Those are not vague complaints. They are consistent with a sensitized nervous system that is no longer filtering properly.

Nerves Becomes Biochemically Unstable
By the time patients develop chronic nerve pain, many assume the nerve must be compressed, damaged, or structurally trapped.
Sometimes that is true. But a lot of the time it is not.
Nerves can also become unstable because of changes in their biochemical environment.
Every nerve relies on tightly regulated ion movement to fire correctly. Sodium, potassium, calcium, chloride, and other ion gradients determine whether a nerve remains quiet, fires appropriately, or becomes hyperexcitable. Neurotransmitters such as GABA help dampen excitability. Local blood flow delivers oxygen and nutrients needed to maintain this balance.
Insulin resistance disrupts several of these stabilizing systems at once. First, it alters ion channel expression. Certain channels involved in sensing inflammation and temperature can become upregulated. That means nerves become more responsive to inflammatory conditions and more likely to generate pain signals.
Second, inhibitory signaling weakens. GABA is one of the nervous system’s primary calming neurotransmitters. If GABA receptor activity is reduced, the braking system weakens. Now excitatory input has less opposition.
Third, membrane stability suffers. Potassium handling is essential for resetting nerve cells after they fire. If potassium regulation becomes impaired, neurons are less able to return to a stable resting state. That makes them easier to re-fire, sometimes with very little provocation.
Fourth, microvascular support declines. Nitric oxide signaling helps regulate blood flow. If nitric oxide signaling drops, circulation to nerves can suffer. Less blood flow means less oxygen, fewer nutrients, worse waste removal, and a greater likelihood of nerve dysfunction.
This is a profound point because it explains why nerve pain can feel so bizarre and so relentless. A biochemically unstable nerve does not need a major trigger. It is already living in an environment that favors reactivity.
So when patients say:
“My nerve pain seems to come out of nowhere”
“It burns even when nothing is touching it”
“It feels electrical for no reason”
“I can’t predict it”
that does not mean the pain is random. Nerves do not fire for no reason. They fire because something is changing the threshold at which they become active. In many people, insulin resistance is part of that threshold problem.
What this means for patients is that nerve pain without diabetes is absolutely possible, because insulin resistance alone can destabilize nerve function before full diabetic disease ever appears.

Why Standard Treatments Often Fail
Most treatments target the end of the pathway:
Anti-inflammatories
Pain medications
Injections
These can temporarily suppress symptoms. But they do not address:
Glucose dysregulation
Insulin signaling
Mitochondrial dysfunction
Nervous system instability
This is why many patients experience diminishing returns over time. The system driving the pain has not been corrected.
Why Your Tests May Look Normal
Many patients are told their labs or imaging are “normal.”
This creates confusion and often self-doubt.
But insulin resistance can exist long before it shows up as diabetes on standard labs. And nervous system dysfunction does not show up on MRI.
Normal tests do not rule out dysfunction. They often just mean the wrong systems are being measured.
The Multi-System Nature of Chronic Pain
Chronic pain is never a single-system problem.
It involves:
Metabolic dysfunction (insulin resistance)
Nervous system sensitization
Immune system dysregulation
Hormonal stress responses
This is why a purely structural or symptom-based approach falls short.
As outlined in the True Health model, chronic pain is always driven by interacting physical, biochemical, and nervous system factors.
Clinical Insight
Many patients come in believing their pain is purely structural—something “wrong” with a joint, disc, or muscle.
But when metabolic dysfunction like insulin resistance is addressed, something unexpected happens:
Pain sensitivity decreases.
Recovery improves.
Flares become less frequent.
This is not because the structure suddenly changed. It’s because the system surrounding that structure became more stable.
You were likely never told that insulin resistance could influence your pain. Not because it isn’t important. But because it does not fit neatly into a single-diagnosis model.
Chronic pain is not a single problem.
It is the output of multiple systems that have lost balance.
When you begin to understand how those systems interact, especially metabolism and the nervous system, your symptoms start to make more sense.
And more importantly, your path forward becomes clearer.

Written By:
Dr. Jason Winkelmann
Naturopathic doctor, Chiropractor, Chronic Pain Specialist, and Educator
Frequently Asked Questions
Do I need diabetes to have insulin-related pain?
No. Insulin resistance can affect nerve function and inflammation long before diabetes develops.
Why does my pain get worse with stress?
Stress increases cortisol, which raises blood glucose and worsens insulin resistance—feeding directly into the pain cycle.
Why do anti-inflammatories stop working?
Because they suppress downstream inflammation without addressing the upstream metabolic dysfunction driving it.
Can insulin resistance affect nerve pain specifically?
Yes. It directly impacts nerve repair, blood flow, ion channel function, and neurotransmitter balance—all critical for nerve health.



Comments